TECHNIQUE COULD TRANSFORM CANCER CARE
Syllabus:
GS-3:
- Science and technology
- Biotechnology
Why in the News?
A new Science study has proposed a method to generate chimeric antigen receptor (CAR) T cells inside the body using targeted lipid nanoparticles and messenger RNA delivery, potentially revolutionising immunotherapy for cancers and autoimmune diseases. This novel in vivo engineering CAR T cell approach avoids lab-based procedures, personalisation costs, and high-dose chemotherapy, making CAR T-cell therapy more accessible and affordable.
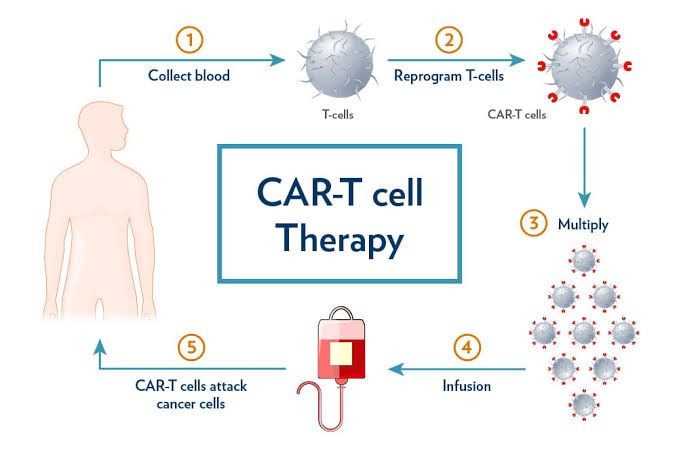
CONCEPT OF CAR-T CELL THERAPY
- Basic idea: CAR T-cell therapy reprograms immune cells to identify and attack rogue targets such as cancerous B-cells using genetically inserted instructions.
- Synthetic boost: Scientists add chimeric antigen receptors (CAR) to T cells, enabling them to recognise proteins like CD19 CAR or CD20 CAR on cancerous cells. This process creates receptor T cells, also known as CAR T cells.
- Post-processing: Modified T cells are reintroduced into the patient to circulate and destroy tumour cells with high precision, a process known as CART therapy.
- High cost: In India, cell CAR T therapy costs ₹60-70 lakh, mostly due to custom lab processing and hospital care.
- Time-consuming: The therapy requires ex vivo manufacturing, chemotherapy for immune suppression, and extensive monitoring for side effects, resulting in a long turnaround time.
NEW METHOD: IN VIVO PROGRAMMING
- Direct delivery: Researchers injected mRNA-loaded lipid nanoparticles (LNPs) directly into immune cells, skipping the need to extract and modify cells externally for CAR T-cell therapy.
- Target specificity: LNPs were tagged with CD8-targeting antibodies to ensure delivery only to killer T cells, increasing precision and safety of the CART therapy.
- Animal trials: In mice and monkeys, these modified T cells attacked B cells, causing tumor clearance and complete B cell depletion.
- Effective results: Up to 85% of T cells in monkeys became CAR-expressing after the second dose, with 95% conversion in associated immune cells.
- Simplified process: This method eliminates ex vivo manufacturing, reduces time, avoids lymphodepletion, and resembles a biological drug infusion.
BYPASSING CURRENT BOTTLENECKS
- No viruses: CARs were delivered via messenger RNA, avoiding permanent genome alterations and risks tied to viral vector usage in traditional CAR T-cell therapy.
- No chemo: Therapy did not require lymphodepleting chemotherapy, avoiding immune suppression and related infections.
- Wider access: The method enables safer administration for elderly or frail patients who can’t endure aggressive treatment.
- Improved carriers: A novel lipid molecule, Lipid 829, ensured low liver toxicity and quick clearance from the body, improving pharmacokinetics and biodistribution.
- Cost-efficient: Without lab engineering or chemo, CART therapy could be dramatically cheaper and more scalable.
APPLICATION IN AUTOIMMUNE DISEASES
- Immune reset: In monkeys, the CAR T treatment led to near-total B cell depletion from tissues like spleen and bone marrow.
- Naive repopulation: Returning B-cells were mostly naive, mimicking healthy immune system reset post CAR T-cell therapy in lupus patients.
- Lupus potential: In vitro tests on systemic lupus erythematosus patient blood showed reprogrammed T cells successfully eliminating harmful B-cells, potentially beneficial for conditions like lupus nephritis.
- Controlled reboot: Transient CAR expression might offer immune reset without chronic immunosuppressive therapy for autoimmune diseases such as myasthenia gravis, systemic sclerosis, and neuromyelitis optica.
- Encouraging data: Though preclinical, the immune reset patterns were consistent with drug-free remission in human trials.
SAFETY AND SIDE EFFECTS
- Usual risks: Conventional CAR therapy causes cytokine release syndrome, neurotoxicity, and secondary infections from low immunoglobulin.
- Lipid benefit: New platform avoided permanent gene edits, reducing risk of long-term side effects on the central nervous system.
- Mild reactions: Inflammation markers like interleukin-6 and interferon-gamma rose slightly but normalised with antihistamines and steroids.
- Tolerable profile: Lipid 829 minimised liver-related effects, showing promise for improved safety in CAR T-cell therapy.
- Rare reaction: One monkey developed a fatal immune overreaction, highlighting the need for careful dosage and close monitoring to prevent cytokine-induced cytotoxicity.
DOSING AND DELIVERY MODEL
- Simple method: Two or three IV infusions spaced 72 hours apart induced strong CAR expression in circulating CD8+ T-cells.
- Drug-like approach: Resembles biologic drug dosing, not lab-intensive cell therapy, with potential for mass standardisation.
- Streamlined care: Avoids customised engineering, reducing infrastructure needs and increasing availability of CAR T treatment.
- Reproducibility challenge: Scaling up from lab trials to industrial settings will require careful standardisation of delivery mechanisms.
- Human trials: Essential to determine efficacy, safety, repeat dosing, and long-term outcomes in patients treated with this new CAR T-cell therapy approach.
RELEVANCE FOR INDIA’S HEALTHCARE
- High burden: India sees large numbers of B cell malignancies, acute lymphoblastic leukaemia, and follicular lymphoma.
- Cost barrier: Traditional CAR T-cell therapy remains unaffordable for most, costing upwards of ₹70 lakh.
- Autoimmune rise: Post-COVID-19, autoimmune cases like multiple sclerosis and idiopathic inflammatory myopathies have risen 30%, boosting demand for affordable CAR immunotherapy.
- Limited access: Few cell therapy units and trained personnel restrict availability, especially in rural regions.
- Wider scope: This model could bring advanced T cell immunotherapy to regions with minimal infrastructure and high patient volumes.
CHALLENGES AND CONCERNS AHEAD
- Human data: Current results are preclinical; safety and efficacy in human trials remain unverified.
- Dosing clarity: Proper dose schedules and cumulative effects must be clearly defined for real-world application of CAR T therapy.
- Regulatory gaps: Need for updated approval frameworks for in vivo therapies using mRNA nanoparticles.
- Manufacturing norms: Reproducibility between R&D labs and automated systems remains a known challenge.
- Long-term monitoring: CAR expression effects on future immune response or organ systems still unknown, including potential resistance mechanisms.
WAY FORWARD FOR POLICYMAKERS
- Pilot trials: Launch controlled trials in India under ICMR to assess feasibility in local populations, including studies on BCMA targeting and CD19 CAR T approaches.
- Price regulation: Introduce guidelines for pricing and insurance coverage to increase patient accessibility to CAR T-cell therapy.
- Tech transfer: Encourage partnerships for technology access and manufacturing LNP platforms in India, including development of CRISPR edited CAR technologies.
- Training boost: Set up dedicated training for oncologists, immunologists, and biotech personnel on in vivo methods and new CAR T approaches.
- Research grants: Allocate funding for translational research on mRNA-based cell therapy in public institutions, including studies on co-stimulation domains and antigen-specific cytotoxicity.
CONCLUSION:
The proposed in vivo CAR T-cell platform using lipid nanoparticles and mRNA may transform cancer and autoimmune disease care by simplifying delivery, reducing costs, and expanding access. This approach could revolutionize treatment for conditions ranging from NALM6 leukemia to hemophagocytic lymphohistiocytosis. With India’s high cancer and autoimmune burden, adopting this approach, following thorough human trials, could redefine CAR immunotherapy in resource-constrained environments. It offers potential for enhancing serial killing capacity of T cells and achieving tumor clearance more efficiently. By addressing challenges like off-target expression and optimizing tissue depletion strategies, this technology could potentially offer curative treatment options for debilitating diseases, bringing hope for drug-free remission in various autoimmune conditions. Future research may focus on genome editing strategies to further improve the efficacy of CAR T-cell therapy and explore its potential in targeting memory B cells responsible for autoantibody production.
SOURCE: TH
MAINS PRACTICE QUESTION:
“Discuss how the recent advances in in vivo CAR T-cell therapy may revolutionise cancer and autoimmune treatment in developing countries like India. Suggest key policy interventions.”








